Vac-Pac Improves the Stability of Argus® SC/ST Salmonella Vaccine
Vac-Pac Improves the Stability of Argus® SC/ST Salmonella VaccineMerck’s Argus® SC/ST is a prominent vaccine manufactured to prevent
the pneumonia, diarrhea, septicemia and mortality in swine caused by Salmonella cholerasuis. It also controls another salmonella species, S. typhimurium, which is one of the common causes of food poisoning in people.
Controlling these infectious bacteria improves swine production efficiency and can reduce the risk of human illness. Salmonella vaccines can provide effective control of salmonella when administered properly. Unlike antibiotics, the avirulent live vaccine fights salmonella by activating the pig’s immune system, allowing the pig to recognize and defeat the more virulent organisms it encounters in its environment. To generate this memory and immunity, the vaccine must be alive when the pig receives it. Argus was developed exclusively for oral dosing through the drinking water delivery system. One of the product’s precautions is to avoid antibiotic use 3 days before or after vaccination, because the antibiotics could kill the vaccine and make it ineffective.
Another risk to the vaccine’s effectiveness is the quality of the drinking water used to deliver the vaccine. Although not included in the Argus precautions, there are other risks that are known to inactivate live vaccines. Particularly threatening to vaccines are oxidizing minerals like iron, nitrate contaminants, or chlorine compounds used to clean the water. Vac-Pac vaccine stabilizer is designed to guard live vaccines against these threats as well as pH and electrolyte imbalances.
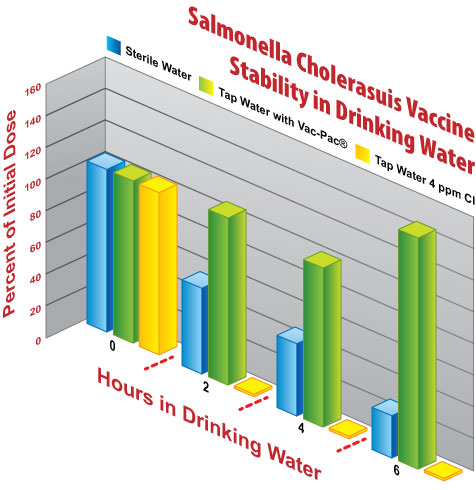
Researchers tested the susceptibility of Argus SC/ST vaccine to oxidation by measuring how long the vaccine survived in three different drinking water sources. The water sources were:
- Sterile distilled water
- Tap water, simulated by chlorinating the sterile distilled water
- The same tap water, stabilized with Vac-Pac
Vaccine was diluted in the drinking water and held for 6-hours. Samples of the water were tested every 2-hours to measure the number of surviving vaccine organisms.
Over the 6-hour time period in sterile distilled water, the vaccine’s potency gradually decayed to 25% of its original dose. Non-stabilized tap water immediately inactivated the vaccine, so that no surviving vaccine organisms could be found at the first time point. Vaccine that had been preserved with Vac-Pac on the other hand maintained all of its original potency throughout the trial.
One key to maintaining successful vaccination is to ensure the animals ingest enough of the organisms to build full immunity. Argus SC/ST vaccine is manufactured and stored in conditions to preserve a target number of freeze-dried organisms in each vial.
Those dehydrated bacteria are dormant, and have a limited life span after they are rehydrated. Vaccines can be quite fragile and susceptible to destruction by harmful elements in the drinking water supply. It becomes clear that, if the vaccine is not properly stabilized, there can be large differences in how much vaccine a pig would receive. The amount of protection lost depends on the conditions of the water and on how long after rehydration the pig consumed the vaccine-containing drinking water.
In contrast, risks to vaccine potency can be reduced by stabilizing the water with Vac-Pac before adding the vaccine. Vac-Pac nurtures the vaccine, rescuing it from decay. It ensures that pigs consume the intended dosage, expressing full immunity and protecting animal health and performance.